Anticoagulant coatings can effectively reduce the risk of thrombosis on the surface of blood-contact medical devices. In the last issue, we introduced two major types of anti-coagulation coating strategies: 'inert' and 'active'. Since different anticoagulant strategies have different mechanisms of action and anticoagulation, the detection methods for pre-evaluation of anticoagulant performance in vitro are also different.
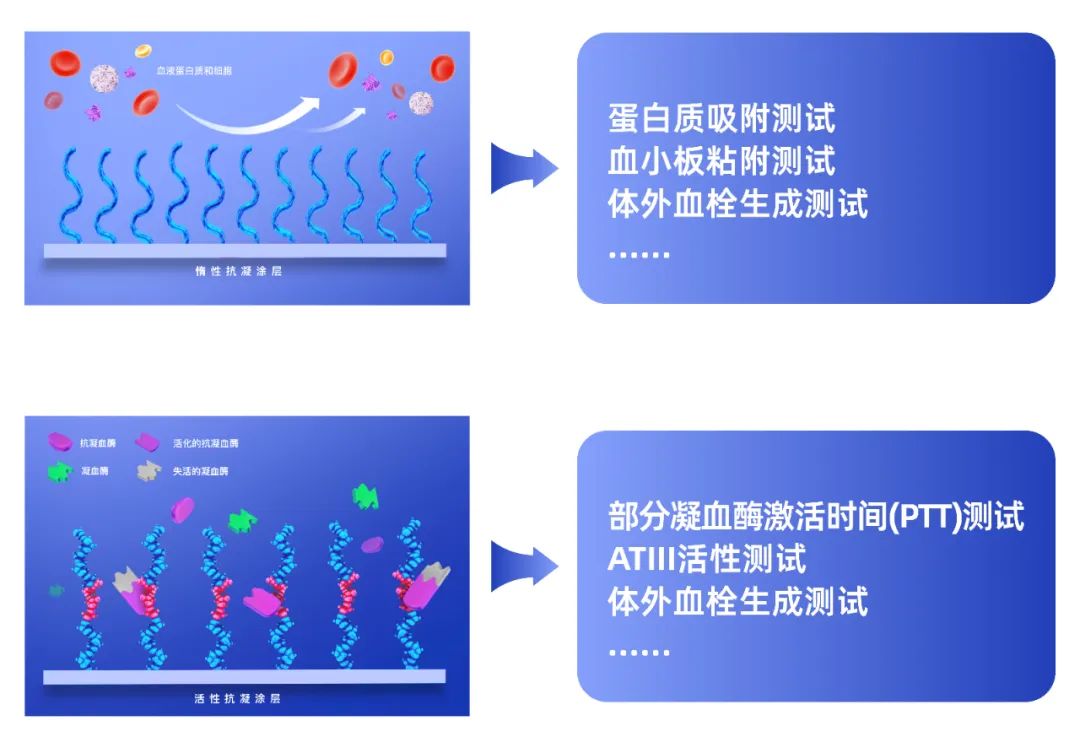
Inert anticoagulant coatings (such as phosphorylcholines) mainly inhibit the activation of the coagulation system by reducing plasma protein adsorption and blood cell adhesion on the material surface. Therefore, protein adsorption tests and platelet adhesion tests are often used in vitro to initially evaluate the anticoagulant properties of inert coatings. Protein adsorption testing usually selects fibrinogen, a key protein in the coagulation system. Testing methods include isotope labeling, fluorescent labeling, eluent concentration measurement (such as BCA method), etc. Platelet adhesion testing can be performed on whole blood or platelet-rich plasma (PRP), followed by observation of morphology and statistical counting by scanning electron microscopy (SEM), or counting by a hematology analyzer.
The most widely used active anticoagulant coating is heparin coating. Heparin inhibits the activity of coagulation factors such as thrombin and Xa by binding and activating antithrombin I (ATIII) in the blood, thereby blocking the coagulation reaction. The direct manifestation of inhibition of coagulation factor activity is the prolongation of coagulation time. Therefore, the most common method for preliminary evaluation of the anticoagulant performance of heparin coating in vitro is to measure the partial thrombin activation time (PTT), or directly measure ATIII activity. In addition, since heparin can be stained by toluidine blue solution, coating integrity, uniformity, and heparin density can be tested by this method.
Of course, no matter which strategy is used, the goal is to ultimately reduce the generation of blood clots on the surface of the product. Therefore, a more intuitive and universal way is to directly observe the amount of blood clots formed on the surface of the product after contact with whole blood. However, this method is not easy to quantify and has certain contingency. Reasonable selection of in vitro performance evaluation methods for anticoagulant coatings during the design and development stage will help to efficiently evaluate the feasibility of the technical route and provide richer scientific supporting data for product registration.

The copyright of this work belongs to Bai Saifei. If you need to reprint it, please contact me.
Tel: (+86) 134 0143 1324
Email: sales@biosurf.cn
Working hours: 08:30-17:30 (Monday - Friday)
Address: Building 26, Dongjing Industrial Park, No. 1 Jintian Road, Suzhou Industrial Park, China
